High-frequency common inputs entrain motoneuron subpopulations differently
High-frequency common inputs entrain motoneuron subpopulations differently
Pascual Valdunciel, A.; Yanguas-Mayo, J.; Abbagnano, E.; Consul, N. T.; Nascimento, F.; Ozyurt, M. G.; Farina, D.; Ibanez, J.
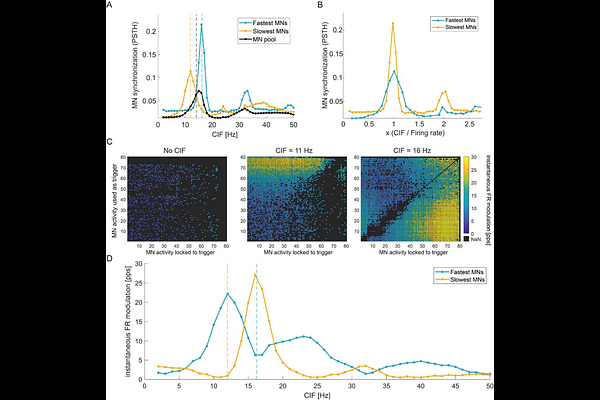
AbstractSpinal motoneuron (MN) pools behave as linear systems that transmit common synaptic input to muscles. However, MNs are biophysically heterogeneous and intrinsically nonlinear. How different MN subpopulations integrate and transmit high-frequency inputs remains poorly understood, partly because conventional analyses treat the MN pool as a single functional system rather than examining subpools with different firing rates. Here, we addressed this gap using a combination of computational simulations and human MN recordings. Simulations of MNs receiving a common synaptic input at varying frequencies showed that MNs' firings become phase-locked to input oscillations when the input frequency approximates the neuron's firing rate or its harmonics. We refer to this frequency-dependent synchronization as entrainment. Importantly, this subpool-specific effect was masked when MN activity was analysed at the whole-pool level. Because entrained MNs effectively sample the input at their firing instants, we developed a MN-firing locked method that uses individual MN firings as endogenous triggering events for peristimulus frequencygrams across the pool. In simulations, this method revealed entrainment-driven firing rate modulations across MN subpools. We then applied this MN-firing locked method to MNs decomposed from high-density surface electromyography recordings obtained during isometric contractions in healthy individuals. We found that faster-firing MNs exhibited larger transient firing rate increases, time-locked to slower MN activity. Furthermore, these modulations correlated with common input in the alpha and beta bands implicating high frequency common input as the driving source. Together, these findings demonstrate that MN nonlinearities generate heterogeneous, frequency-dependent dynamics that remain hidden in conventional pool-level analyses.