A Nickel N-Heterocyclic Biscarbene Complex Derived from Caffeine Enhances Fluconazole Efficacy against Candida glabrata
A Nickel N-Heterocyclic Biscarbene Complex Derived from Caffeine Enhances Fluconazole Efficacy against Candida glabrata
Malta-Luis, C.; Romeo, G.; Francescato, G.; Mariano, C.; Mil-Homens, D.; Petronilho, A.; Pimentel, C.
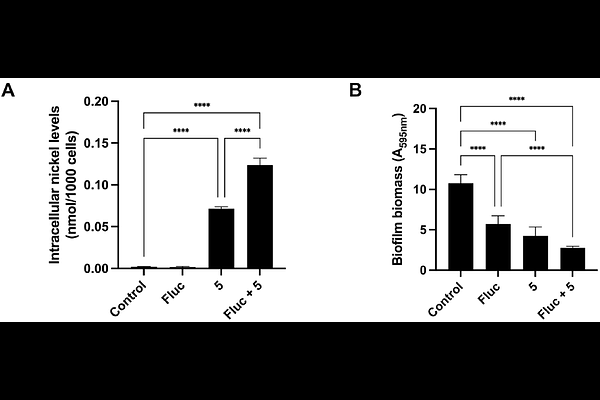
AbstractInvasive infections caused by Candida spp. are associated with high morbidity and mortality, particularly in immunocompromised patients, and are increasingly difficult to treat due to rising antifungal resistance. Here, we further investigate the antifungal properties of a previously reported nickel N-heterocyclic biscarbene derived from caffeine (compound 5) and describe the synthesis of an analogous nickel N-heterocyclic biscarbene based on benzimidazole (compound 8), designed to evaluate the impact of the biscarbene and xanthine frameworks on activity and toxicity. Compound 5 displayed selective activity against Candida glabrata, inhibited biofilm formation and showed greater cellular accumulation in this species. In a Galleria mellonella infection model, 5 significantly reduced fungal burden while exhibiting lower cytotoxicity than the benzimidazole analogue 8. Importantly, although compound 5 and fluconazole are individually fungistatic, their combination was fungicidal against C. glabrata. In the presence of compound 5, the minimal inhibitory concentration of fluconazole decreased against both a fluconazole resistant petite mutant and a respiratory competent C. glabrata strain evolved in vitro to fluconazole resistance. Compound 5 increased the frequency of petite mutants, suggesting an effect on mitochondrial function; however, its retained activity against these mutants and its synergism with fluconazole in the petite background indicate an additional mechanism of action. These findings identify 5 as a promising antifungal adjuvant and support its potential use in combination therapy to enhance azole efficacy against C. glabrata.