Activation of the protective arm of renin-angiotensin system enhances mitochondrial turnover improving respiration and decreasing integrated stress response in a human Complex III deficiency model.
Activation of the protective arm of renin-angiotensin system enhances mitochondrial turnover improving respiration and decreasing integrated stress response in a human Complex III deficiency model.
Fernandez-Del-Rio, L.; Eastes, A.; Rincon Fernandez-Pacheco, D.; Scillitani, N.; Garza, J.; Dugan, M.; Pinto de Oliveira, M.; Kadam, P.; Gauhar, I.; Erion, K.; Rodgers, K.; Gaffney, K.; Wang, A.; Liesa, M.; Beninca, C.; Shaul Shirihai, O.
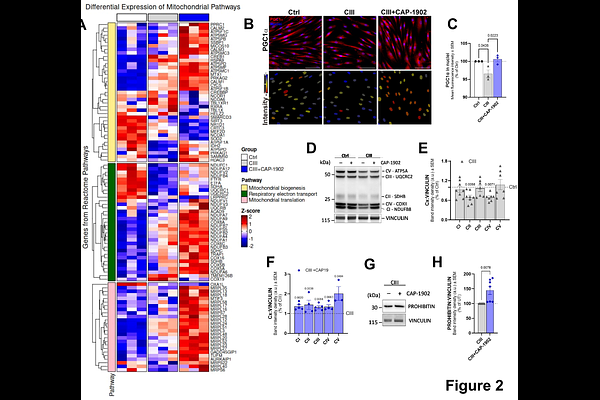
AbstractPrimary mitochondrial diseases are clinically and genetically heterogeneous disorders, commonly caused by defects in the oxidative phosphorylation system. This heterogeneity presents major challenges for therapeutic development; however, a shared hallmark across these diseases is the accumulation of dysfunctional mitochondria. Enhancing mitochondrial turnover, by activating the selective degradation of dysfunctional mitochondria via mitophagy, concurrently with the activation of mitochondrial biogenesis, could represent a shared therapeutic strategy for mitochondrial diseases. Here, we describe a novel mitophagy inducer, CAP-1902. CAP-1902 is a new agonist of the MAS G-Protein Coupled Receptor (MasR). In fibroblasts from patients carrying a BCS1L mutation that impairs complex III (CIII) assembly, CAP-1902 increased mitochondrial turnover by promoting both mitophagy and biogenesis. Specifically, MasR activation triggered the AMPK/ULK1/FUNDC1 mitophagy pathway. Knockdown of FUNDC1 blocked mitophagy but not AMPK activation, confirming pathway specificity. Additionally, a decrease in the occurrence of depolarized mitochondria with treatment indicated the selective targeting of accumulated damaged mitochondria in the disease context. MasR activation by CAP-1902 also stimulated the nuclear translocation of PGC-1, promoting increased expression of transcripts associated with mitochondrial biogenesis, respiratory chain components, and mitochondrial translation. Remarkably, CAP 1902 was ultimately able to restore key defects in CIII-deficient fibroblasts by rescuing bioenergetics and correcting both the aberrant lysosomal distribution and the elevated integrated stress response markers, which is consistent with a shift toward a healthier mitochondrial population. In summary, we describe the first potential GPCR-mediated treatment of mitochondrial diseases and demonstrate that MasR activation by CAP-1902 induces mitochondrial turnover and improves mitochondrial function.