Accessing pore-blocker bound and open conformations of TMEM16A using PIP2-assisted adaptive sampling
Accessing pore-blocker bound and open conformations of TMEM16A using PIP2-assisted adaptive sampling
Pipatpolkai, T.; Yong, E. H.
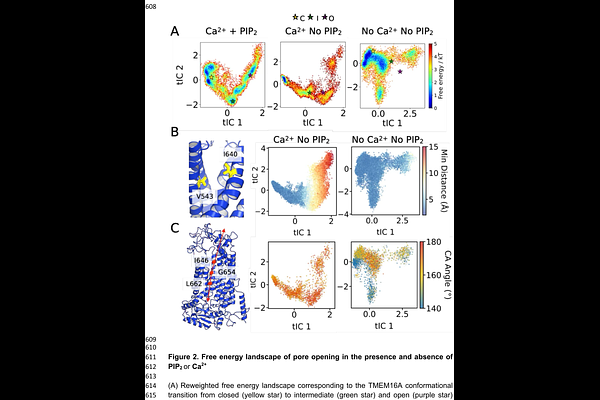
AbstractIon channels are promising targets for drug discovery due to their diverse physiological functions. The database of ion channel structures has grown exponentially over the last decade due to advances in structure-determination techniques. However, not all ion channel conformations have been determined, and not all druggable conformations can be modelled as thermodynamically stable under simulation conditions. This greatly limits conformation-specific drug targeting. In this study, we used an endogenous regulator of ion channels, phosphatidylinositol-4,5-bisphosphate (PIP2), as a computational tool to probe the open-state conformation of the TMEM16A calcium-activated chloride channel. By using the PIP2-binding conformation from a coarse-grained model, followed by fluctuation-amplified specific traits (FAST) adaptive sampling in an all-atom configuration, the system transitioned from a closed state to a thermodynamically stable open state. The transition also highlights the importance of PIP2 in TM6 helical kink, the opening of the outer gate and the alpha helix on I551. The open-state structure displays the experimental conductance. Using an accelerated weighted histogram (AWH), the binding site of 1PBC, A9C, niclosamide and Ani9 pore blockers were determined and validated against previous experimental studies. This paves the way to structure-specific drug development, as overactivation of TMEM16A is correlated with many diseases, such as pulmonary hypotension and ischemic stroke. Together, this study highlights the importance of lipids in stabilising ion channel conformations for targeted drug design and introduces a novel approach to expand the therapeutic targeting of ion channels.