Lytic bacteriophages active in urine against multi-drug resistant clinically derived Klebsiella pneumoniae causing urinary tract infection
Lytic bacteriophages active in urine against multi-drug resistant clinically derived Klebsiella pneumoniae causing urinary tract infection
Calin, R.; Bernabeu Vilaplana, B.; Gedeon, J.; Capton, E.; Galinat, C.; Saffarian, A.; Pierrat, G.; Benzerara, Y.; Wurtzer, S.; Moulin, L.; Eckert, C.; Tournebize, R.
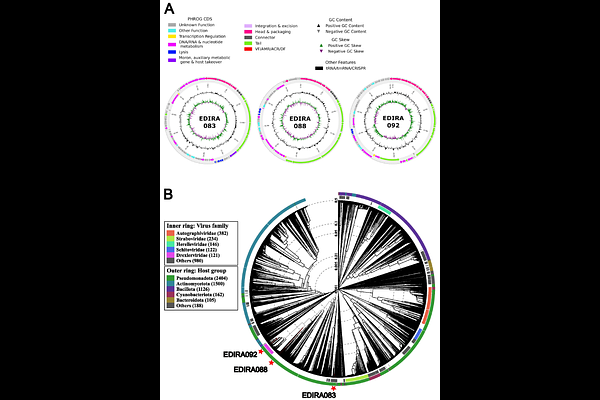
AbstractObjectives Multidrug-resistant (MDR) Klebsiella pneumoniae is an increasingly important cause of recurrent urinary tract infections (UTIs), particularly in high-risk patients such as those with neurogenic bladder, where therapeutic options are limited. Bacteriophage therapy represents a promising alternative, but pre-clinical models and characterization of phages active against UTI-derived strains remain scarce. We therefore aimed to isolate and characterize bacteriophages targeting a clinical MDR K. pneumoniae strain causing recurrent UTI and evaluate their activity under urinary conditions. Methods Three bacteriophages were isolated from environmental samples using an ESBL-producing K. pneumoniae clinical isolate obtained from a neurogenic bladder patient. Phages were characterized by genome sequencing, electron microscopy, stability assays, one-step growth curves, and host-range analysis across 79 clinical UTI isolates. Phage activity was quantified in LB medium and human urine using bacterial growth kinetics and a lytic activity score. Results Three lytic phages from the former siphoviridae family (EDIRA083, EDIRA088, and EDIRA092) belonging to distinct genera were identified. Genomic analysis confirmed the absence of lysogeny-associated, virulence, or antibiotic-resistance genes. Latent periods ranged from 8 to 40 minutes and burst sizes from 38 to 170 virions per infected bacterium. Host-range analysis revealed narrow activity for EDIRA083 and EDIRA088, whereas EDIRA092 infected 29% of the 79 clinical isolates tested. In liquid phage infection assays, overall lytic activity was consistently higher and more sustained in human urine than in LB, suggesting reduced fitness of resistant mutants under urinary conditions. Conclusions These results identify three genetically distinct lytic phages targeting MDR K. pneumoniae and highlight the importance of testing phage activity under infection-relevant conditions. Their activity in urine supports further evaluation of these phages as candidates for therapeutic development against MDR Klebsiella UTI.