Cryo-EM structures of mycobacterial MCC reveal carrier-domain translocation between catalytic sites
Cryo-EM structures of mycobacterial MCC reveal carrier-domain translocation between catalytic sites
Yadav, A.; Florea, B. I. I.; Geibel, S.
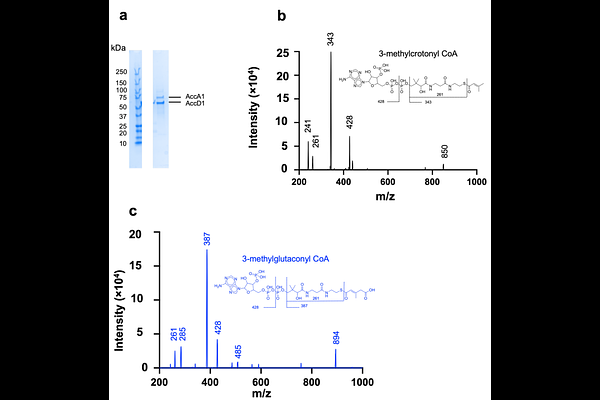
Abstract3-Methylcrotonyl-CoA carboxylase (MCC) is a biotin-dependent enzyme complex that catalyzes an essential step in leucine degradation. In mycobacteria, MCC is formed by AccA1 and AccD1, but its structural organization has remained poorly defined. Here, we report two high-resolution cryo-EM structures of the endogenous, biotin-bound {beta} complex from Mycobacterium smegmatis. The MCC architecture comprises a central hexameric carboxyltransferase ({beta}/CT) core flanked by trimeric biotin carboxylase (/BC) modules. The structures capture BC- and CT-engaged conformations, in which the biotin carboxyl carrier protein (BCCP) engages either the BC or CT active site, providing structural evidence for long-range carrier-domain translocation. Conformational rearrangements within the BC module suggest a role in regulating BCCP engagement. Together, our findings provide insights into carrier-domain dynamics in mycobacterial MCC