Ovary-Derived Signals Align Protein Appetite with Oogenesis
Ovary-Derived Signals Align Protein Appetite with Oogenesis
Nobrega, R. R.; Francisco, A. P.; Gontijo, A. M.; Ribeiro, C.; Carvalho-Santos, Z.
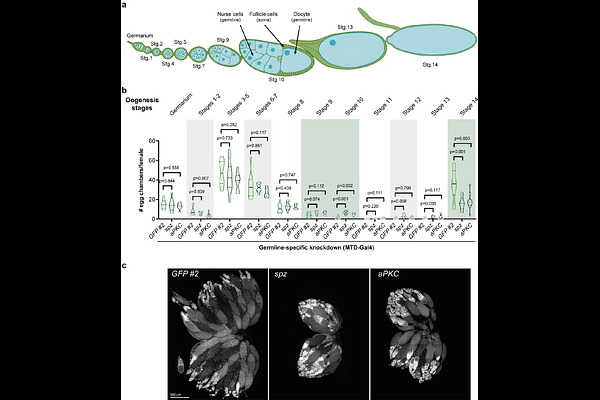
AbstractMaintaining organismal homeostasis requires mechanisms that coordinate the metabolic needs of individual organs with whole animal nutrient intake. Although nutrient sensors and central pathways regulating hunger have been extensively characterized, how peripheral organ physiology influences nutrient specific appetites remains poorly understood. Here, we identify a previously unrecognized signalling axis, originated in the ovary, that modulates yeast appetite in Drosophila melanogaster. Through a targeted germline RNAi screen, we find that specific perturbations in oogenesis consistently and selectively increase yeast appetite. The manipulations increasing yeast appetite disrupt oogenesis progression, producing a shared signature of increased vitellogenic follicle accumulation and reduced number of mature (stage14) oocytes. This shift is accompanied by decreased expression of the relaxin-like hormone Dilp8, and loss of Dilp8 recapitulates the feeding phenotype. We show that this ovarian regulation of nutrient specific appetite is independent of amino acid state but requires mating, indicating integration with Sex Peptide-mediated reproductive activation. Together, our findings uncover a novel mechanism that couples oogenesis progression to nutrient selection, emphasizing the importance of the ovary as an active regulator of whole organism nutritional decisions. This work provides a conceptual framework for how reproductive tissues communicate their physiological demands to other organs and raises the possibility that analogous ovary-derived signals may shape nutrient specific appetite and metabolic states in other animals.