Mitochondrial malate metabolism acts as a control hub for photosynthesis and carbon-nitrogen balance in Arabidopsis
Mitochondrial malate metabolism acts as a control hub for photosynthesis and carbon-nitrogen balance in Arabidopsis
Martinez, M. d. P.; Nica, I.; Zheng, K.; Ditz, N.; de Oliveira, J. A. V. S.; Barreto, P.; Blum, N.; Westhoff, P.; Pucker, B.; Eubel, H.; Finkemeier, I.; Scharzlaender, M.; Maurino, V. G.
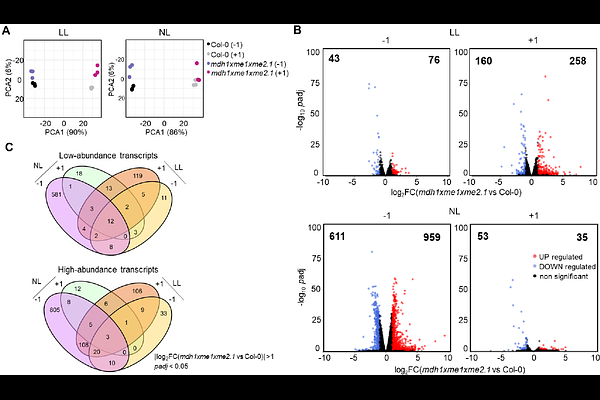
AbstractMalate is a central metabolite in plant energy metabolism and biosynthesis and serves as a major carrier of carbon and reducing equivalents between chloroplasts, the cytosol, and mitochondria. However, how individual malate-converting systems contribute to physiology in specific subcellular compartments remains incompletely understood. Here, we investigated the impact of combined loss of mitochondrial malate dehydrogenase (MDH) and NAD-dependent malic enzyme (NAD-ME) activity in Arabidopsis thaliana by integrating reverse genetics, physiological analyses, transcriptomics, quantitative proteomics, and metabolite profiling. Specifically, we generated triple mutants (mdh1xme1xme2) lacking the predominant mitochondrial isoform MDH1 together with both NAD-ME subunits, thereby reducing overall mitochondrial malate conversion capacity. By growing plants under contrasting photoperiod and irradiance regimes to vary photosynthetic demand on malate-linked fluxes, we uncovered a conditional phenotype that was most pronounced under short-day/low-light conditions. Under these conditions, mdh1xme1xme2 exhibited impaired growth and photosynthetic performance, accompanied by cytosolic redox imbalance and altered chloroplast ultrastructure. Transcriptomic profiling revealed that low light unmasks a dawn-phase bottleneck in establishing photosynthetic and redox homeostasis. Consistent with this, the low-light plastid proteome revealed a reallocation away from chloroplast translation and photosynthetic capacity toward proteome maintenance, photoprotection/repair, and iron/ROS management, consistent with a protective acclimation state that nevertheless constrains carbon gain under energy limitation. Low light also triggered C/N imbalance and ammonium accumulation in the mutants. In contrast, increasing irradiance or extending the photoperiod largely alleviated these defects. Together, our results identified mitochondrial malate conversion capacity as a key control point coupling respiratory energy supply and redox homeostasis to photosynthetic metabolism when photosynthetic energy input is limiting.