Structural insight into sodium-dependent bile acid transport by members of the SLC10 family
Structural insight into sodium-dependent bile acid transport by members of the SLC10 family
Li, C. Y.; Grob, A.; Repa, L.; Huxley, O.; Brotherton, D. H.; Becker, P.; Dadzie, R.; Beckstein, O.; Cameron, A. D.
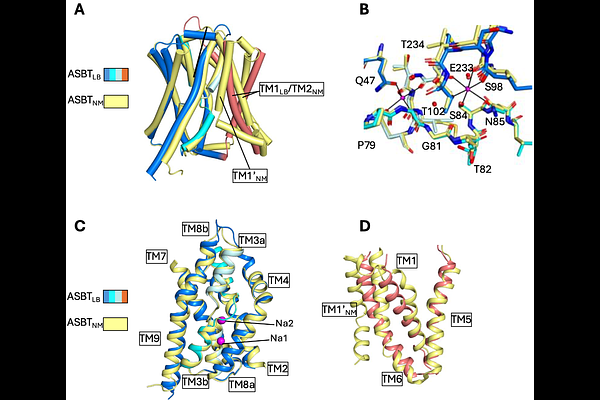
AbstractASBT and NTCP are sodium-coupled secondary transporters of the SLC10 family that play critical roles in the enterohepatic recycling of bile acids. Secondary transporters generally function through the alternating access mechanism. However, in outward-facing structures of NTCP this mechanism is violated with a pore running through the protein. No such pore is observed in previously reported structures of bacterial ASBT homologues, but these proteins have an additional transmembrane-helix adjacent to the binding site. Here we investigate another homologue of ASBT without this extra helix. We solve the crystal structure of the inward-facing state in multiple conformations and a humanised version in the outward-facing state, with and without bile acid bound. Unlike for NTCP there is no pore through the protein as the exit is blocked by the flexible TM6. Consequently, the presence of the pore is not necessarily common to the family. Molecular dynamics simulations of both bacterial and human proteins highlight that while the bile acid substrate is anchored to residues at the centre of the transporter, lipids from the surrounding membrane interact with the hydrophobic sterol group. Incorporating lipids into the transport mechanism can explain how bile acids modified by large hydrophobic groups can bind and be transported.