Structural and cellular insights into the inhibition of the drug efflux activity of the HEDGEHOG receptor PATCHED1
Structural and cellular insights into the inhibition of the drug efflux activity of the HEDGEHOG receptor PATCHED1
Houha, O.; Wachich, M.; Debarnot, C.; Kovachka, S.; Azoulay, S.; Mus-Veteau, I.; Biou, V.
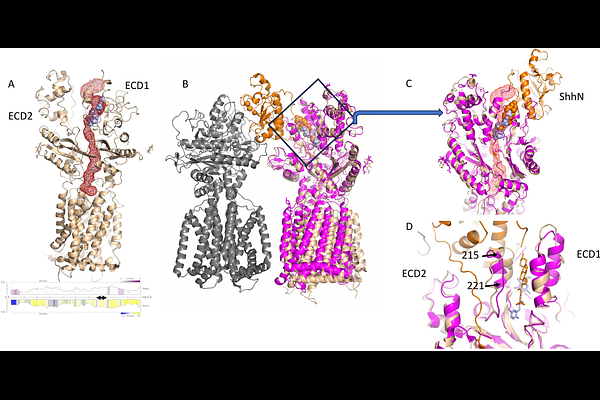
AbstractPATCHED1, the receptor for the Sonic Hedgehog morphogen, mediates cholesterol transport across the plasma membrane by harnessing the proton motive force. In cancer, PATCHED1 is frequently overexpressed and promotes chemoresistance by transporting drugs such as doxorubicin (dxr) out of cells. Among the inhibitors identified, PAH stands out for its ability to significantly enhance dxr efficacy across various cell types, including cancer cells. To investigate the role of PATCHED1, we overexpressed a construct spanning residues 1-619 and 720-1305 in HEK293 cells. The protein localized to the membrane, and transfected cells exhibited reduced sensitivity to dxr compared to controls. Additionally, we observed a pH-dependent decrease in intracellular dxr fluorescence, which was reversed by PAH, confirming that the PATCHED1 construct functions as an active drug-efflux pump. Using cryo-electron microscopy, we determined the structure of PATCHED1 bound to PAH. PAH occupies a hydrophobic cavity in an extracellular domain - normally occupied by cholesterol in other PATCHED1 structures - and engages in a key hydrogen bond via one of its hydroxyl groups, a feature previously established as vital for its inhibitory function. Notably, a second hydroxyl group resides in a more spacious cavity, presenting an ideal site for introducing bulkier chemical modifications. These findings not only clarify the molecular basis of PAH action but also provide a structural roadmap for rational drug design, enabling the development of next-generation inhibitors with enhanced potency.