Human breast milk extracellular vesicles from mothers with asthma differentially modulate the release of inflammatory cytokines by primary human airway smooth muscle cells in a recipient-cell specific manner
Human breast milk extracellular vesicles from mothers with asthma differentially modulate the release of inflammatory cytokines by primary human airway smooth muscle cells in a recipient-cell specific manner
Souza, T. F.; Pierdona, T. M.; Seif, S.; Bydak, B.; Obi, P. O.; Gordon, J. W.; Turvey, S.; Simons, E.; Mandhane, P.; Moraes, T.; Subbarao, P.; Raghavan, S. A.; Halayko, A. J.; Azad, M. B.; Saleem, A.
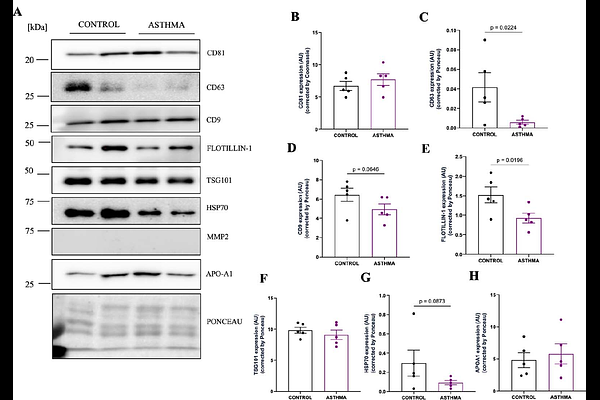
AbstractBreastfeeding provides health benefits in childhood, reducing the frequency of gastrointestinal and respiratory infections. Breastmilk (BM) is a rich source of bioactive molecules including extracellular vesicles (EVs), which exert immunomodulatory signalling in recipient cells, with cargo that is affected by maternal characteristics. Here we investigated the biophysical characteristics of BM-EVs from mothers with (asthmatic BM-EVs) or without asthma (control BM-EVs) and their effect on the release of cytokines from primary human hTERT-immortalized airway smooth muscle cells (hASMs) from asthmatic or non-asthmatic (control) donors. BM-EVs were isolated using size exclusion chromatography (N=5/group), characterized biophysically and by EV-specific protein markers. In addition, BM-EV were co-cultured (48h) with primary hASM cells from both non-asthmatic (control) and asthmatic donors to determine the effect on cytokine release. All participants were Caucasian and the BM was collected 12-15 weeks postpartum. BM-EVs showed the presence of intact and small-EVs (~100 nm). Asthmatic BM-EVs appeared to have a smaller average EV size (135.6 nm) vs. controls (148.3 nm, p=0.0613), but ~5-fold higher concentration of both total (p=0.0014) and small EVs (p=0.0016). The expression of EV subtype protein expression was reduced in asthmatic BM-EVs vs. control BM-EVs: CD63 by 86% (p=0.0224), flotillin-1 by 40% (p=0.0196), CD9 by 24% (p=0.0646) and HSP70 by 69% (p=0.0873). Asthmatic BM-EVs co-cultured with hASMs from control donors decreased pro-inflammatory cytokine release: MCP-1 by 55% (p=0.0286), IL-6 by 45% (p=0.0801) and IL-2 by 32% (p=0.0970) vs. control-BM-EVs. Conversely, asthmatic BM-EVs co-cultured with hASMs from asthmatic donors increased secretion of anti-inflammatory cytokine IL-10 by 32% (p=0.0660), and IL-1Ra by 75% (p=0.0875), and pro-inflammatory IL-2 by 57% (p=0.0688) vs. control-BM-EVs. Internalization of control and asthmatic BM-EVs was confirmed by labelled EV uptake experiments. No detrimental effects on cell viability with BM-EV treatment were observed. In summary, asthmatic BM-EVs are smaller and enriched in BM, and exert differential effects on cytokine release in a BM-donor and recipient-cell specific manner. Given that BM can enter infant airways, the immunomodulatory effects of BM-EVs on hASMs warrants further investigation to delineate the under underlying mechanisms.