Early postnatal Flt3+ hematopoietic progenitors realize fate-restricted and long-lived output in vivo
Early postnatal Flt3+ hematopoietic progenitors realize fate-restricted and long-lived output in vivo
Cirovic, B.; Nizharadze, T.; Dietlein, N.; Henrich-Kellner, C.; Hoefer, T.; Rodewald, H.-R.
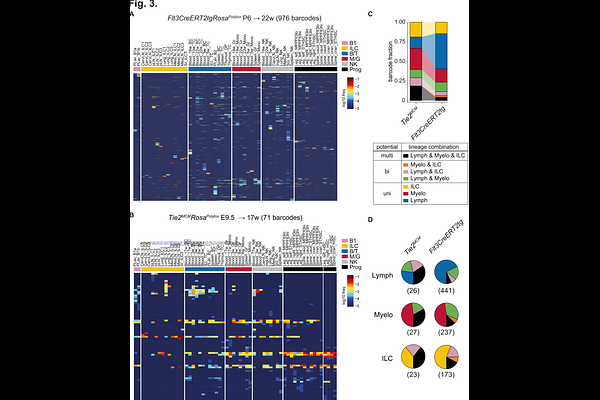
AbstractHematopoietic progenitors downstream of hematopoietic stem cell (HSC) are now recognized as the main drivers of day-to-day hematopoiesis. While embryonic and adult HSC fates have been studied in detail, less information exists on stages downstream from HSC, notably in the multipotent progenitor compartment. The early postnatal period represents an important growth phase of the animal and its immune system. Developing immune lineages must be generated in large numbers rapidly, and populate expanding organ niches. To shed light on this critical period, we focused our experiments on early postnatal Flt3+ hematopoietic progenitors, and combined genetic single progenitor barcoding using Polylox with Flt3-driven, inducible fate mapping. Key immune cell types, including T and B lymphocytes (lymphocytes), innate lymphocytes (ILC) 1-3, NK cells, and granulocytes and monocytes (myeloid) emerged from Flt3+ hematopoietic progenitors. Barcode analysis revealed that about 75% of Flt3+ hematopoietic progenitors had unipotent fates for lymphocytes, or ILC or myeloid cells, while the remaining fraction showed unprecedented fate combinations for these lineages. Focusing on ILC only, we uncovered clonal fate restriction towards ILC1, or ILC2, or ILC3 in tissues. These data indicate early tissue seeding by progenitors, and further differentiation towards discrete subsets in situ. In addition to these fate analyses, induction of fluorescent marker at this intermediate stage of hematopoiesis showed that Flt3+ progenitors generated a wave of progeny lasting for over one year. The washout of these cells over time provided kinetic data of cell turnover in major immune cell compartments (in the circulation and in tissues) in vivo. In conclusion, we tracked the fate of large numbers (in the order of hundreds) of Flt3+ progenitor clones in situ. These intermediate progenitors downstream of HSC displayed mostly lineage-restricted fates as well as strong fate complexity, thus serving as a source for early tissue seeding and durable immune lineage.