In Cellulo pharmacological profiling and genomic editing reveals paralog-specific targets for PA generation during PLC signaling
In Cellulo pharmacological profiling and genomic editing reveals paralog-specific targets for PA generation during PLC signaling
Weckerly, C. C.; Murtagh, O. L.; Swayhoover, T.; Pemberton, J.; Hsu, K.-L.; Hammond, G. R.
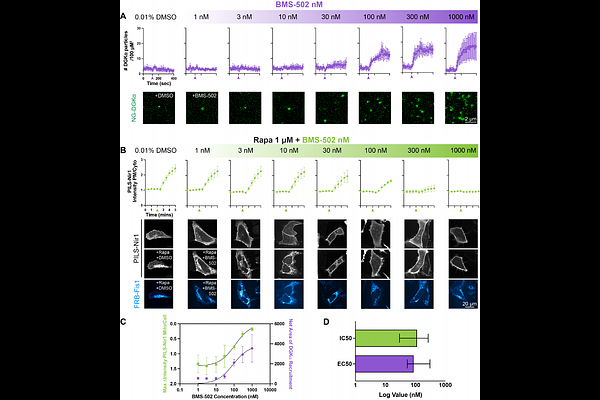
AbstractPhosphatidic acid (PA) is an essential intermediate generated during phospholipase C (PLC) signaling, but its regulation is complex. PA can be generated by ten different diacylglycerol kinase paralogs (DGKs) and two different phospholipase D paralogs (PLDs) in mammals. Because these enzymes are activated under diverse conditions and at various membranes, understanding paralog-specific contributions to PA production is critical for therapeutic development of drugs that modulate the PLC pathway. To address this, we aimed to characterize the paralog specificity of the DGK inhibitors R59022 and BMS-502 against individual DGK paralogs in cellulo. We found that R59022 and BMS-502 both recruited endogenous DGKalpha to the plasma membrane, and inhibited the catalytic fragment of DGKalpha when ectopically localized to the mitochondrial outer membrane. However, at its effective dose, R59022 paradoxically increased PA levels and was cytotoxic, while BMS-502 functioned as a potent and nontoxic inhibitor. Live-cell imaging experiments using BMS-502 with carbachol stimulation of endogenous muscarinic receptors showed that inhibition of both DGKalpha and the PLDs is needed to substantially reduce PA levels during PLC activation. Our findings both identify paralog-specific druggable targets for modulating PLC signaling events, and establish a new platform that translates typical biochemical dose response assays in cellulo.