Secretion and signaling properties of Wnt11 and Wnt3 are determined by their N-terminus
Secretion and signaling properties of Wnt11 and Wnt3 are determined by their N-terminus
Voloshanenko, O.; Dhonnar, N.; Aponte-Santamaria, D.; Seidl, C.; Wolf, L.; Kranz, D.; Ivanova, A.; Figueroa Marquez, E. J.; Pokrandt, B.; Augustin, I.; Port, F.; Sinning, I.; Niehrs, C.; Bruegger, B.; Boutros, M.
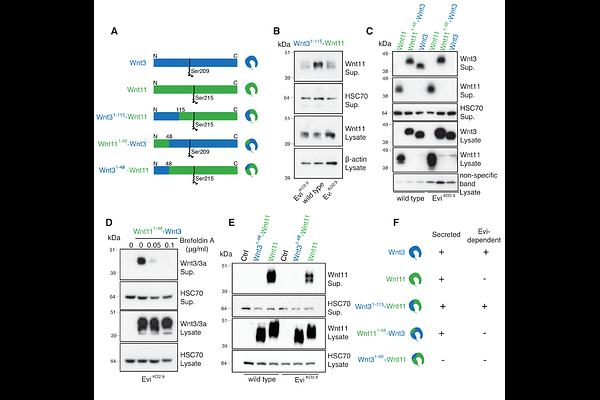
AbstractWnt proteins are evolutionarily conserved signaling molecules that control cell-cell communication in health and disease. While different Wnt proteins share a requirement for palmitoleoylation and the cargo receptor Evi/Wls for their secretion, they interact with diverse signaling receptors and co-receptors to elicit distinct outcomes in signal-receiving cells. The molecular mechanisms underlying this contrast between conserved post-translational modification and secretion and diverse receptor interactions remain poorly understood. Here, we demonstrate that Wnt11 maintains partial secretion capability in the absence of Evi/Wls. Unexpectedly, the secreted protein is also non-palmitoleoylated, yet retains its ability to activate downstream signaling in receiving cells through the Frizzled 6 receptor. We identify the N-terminus as the mediator of this unusual behavior, as Wnt3, which strictly requires Evi/Wls, becomes Evi/Wls-independent when its N-terminus is replaced with that of Wnt11. In different cell lines and in Xenopus laevis embryos, the chimeric Wnt11Nterm-Wnt3 protein recapitulates known Wnt11 phenotypes, demonstrating that the N-terminal portion of Wnt11 is sufficient to determine signaling specificity. Our findings reveal novel determinants of Wnt protein specificity and provide mechanistic insights into the molecular basis of Wnt secretion and downstream signaling.