Ca2+ influx through ER-plasma membrane contacts is required for brown fat thermogenesis and metabolic health
Ca2+ influx through ER-plasma membrane contacts is required for brown fat thermogenesis and metabolic health
Zhou, J.; Dogan, C.; Artico, L. L.; Rodrigues Santos, K.; Hakam, S.; Kim, T.; Xu, V.; Lapenta, K.; Kang, M.; Jorgens, D. M.; Widenmaier, S.; Parlakgul, G.; Arruda, A. P.
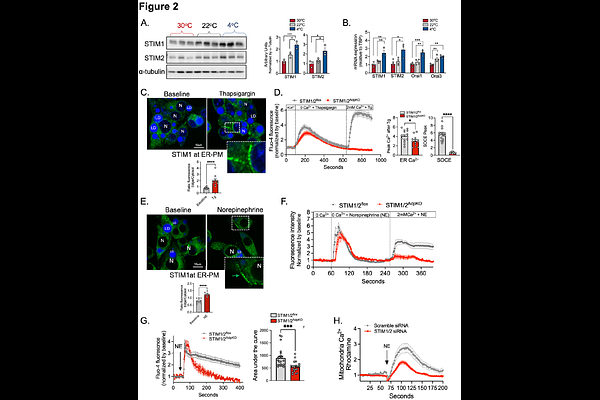
AbstractBrown adipose tissue (BAT) exhibits exceptional metabolic plasticity, rapidly increasing energy expenditure to sustain thermogenesis during cold exposure. This high metabolic activity imposes substantial demands on cellular systems, requiring robust adaptive mechanisms to maintain homeostasis and prevent cellular stress. Yet, the pathways that support and coordinate these adaptive responses in brown adipocytes remain incompletely understood. Here, we identify a cold-induced adaptive program in BAT characterized by the formation of endoplasmic reticulum-plasma membrane (ER-PM) contact sites and the activation of store-operated calcium entry (SOCE), which is essential for maintaining brown adipocyte health during thermogenic activation. Cold exposure enhances ER-PM contacts and upregulates the expression of STIM and Orai proteins, key mediators of SOCE. Loss of STIM in brown adipocytes disrupts intracellular calcium homeostasis and induces aberrant aggregation of ER membranes. STIM deficiency also impairs cold-induced mitochondrial fission resulting in hyperfused mitochondria with reduced oxidative capacity, independently of UCP1 abundance. Importantly, mice lacking STIM in BAT exhibit impaired lipid oxidation, are cold intolerant and develop exacerbated peripheral insulin resistance when challenged with a high-fat diet. Together, these findings identify ER-PM remodeling and STIM-mediated SOCE as a central regulator that links organelle architecture to brown adipocyte function and contributes to whole-body metabolic homeostasis.