Müller glia subtypes define neuro-glial associations and spatial morphogen axes in the zebrafish retina
Müller glia subtypes define neuro-glial associations and spatial morphogen axes in the zebrafish retina
Storey, S. S.; Hehr, C. L.; Standing, S.; McFarlane, S.
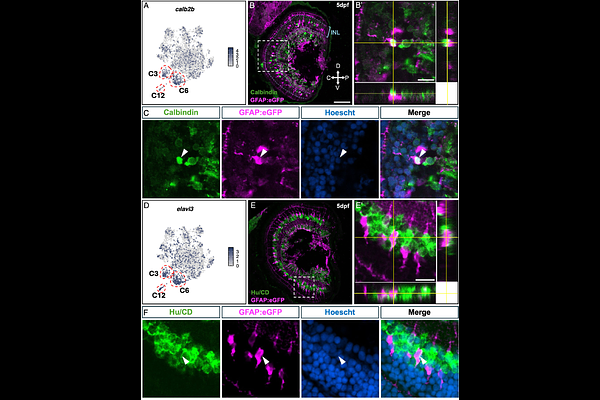
AbstractMuller glia are instrumental macroglia of the vertebrate retina, once thought to be a homogeneous population. Now Muller glia are generally accepted as transcriptionally heterogeneous, and new evidence suggests functional diversity may exist in the way these cells respond to retinal injury. It remains unclear, however, whether this functional heterogeneity is limited to a transient phenotype that stems from injury or a fundamental feature of the healthy retina. Here, we investigate Muller glia heterogeneity in the uninjured zebrafish retina across development and adulthood using a comprehensive single-cell transcriptomic atlas of the 5 days post-fertilization (dpf) eye, validated in vivo and integrated with 9 dpf and adult datasets. We reveal that Muller glia are partitioned into three constitutive subpopulations that persist from early larval stages into adulthood: 1) a proliferative and immature population in both the peripheral and central retina; 2) a novel cohort of neuron-associated Muller glia that express coherent transcriptional programs specific to distinct neuronal subtypes, including retinal ganglion, amacrine and horizontal cells; and 3) spatially distinct Muller glia subsets that define a dorso-ventral axis of retinoic acid metabolism, bisected by a novel cyp26c1-expressing equatorial domain. Finally, cross-species analysis reveals that while neuron-associated programs are evolutionarily conserved in mammals, the spatial patterning of morphogens in adult retinae may be specific to the teleost lineage. Collectively, these findings provide robust evidence for intrinsic functional heterogeneity in the uninjured vertebrate retina, reframing Muller glia from a general support population to a specialized cellular network that actively maintains retinal geography and function.