Best practices for cryo-trapping time-resolvedcrystallography with the Spitrobot crystal plunger
Best practices for cryo-trapping time-resolvedcrystallography with the Spitrobot crystal plunger
Bosman, R.; Hatton, C. E.; Prester, A.; Spiliopoulou, M. E.; Tellkamp, F.; Mehrabi, P.; Schulz, E. C.
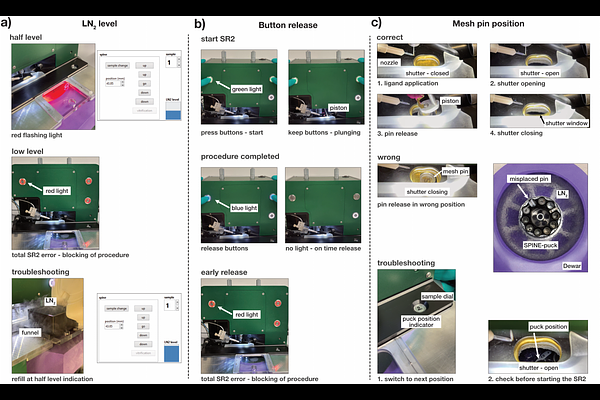
AbstractCapturing meta-stable conformations of enzymes and ligand complexes demands structural snapshots beyond static crystal structures. While time resolved serial crystallography at room temperature, offers a time-resolution down to the femto-second domain it requires large amounts of micro crystals, specialized beamlines and considerable experience. Moreover, as the majority of enzymes displays turnover-times in the millisecond domain or slower, simpler methods can provide meaningful structural insight into enzyme catalysis. Vitrification of protein crystals can trap reaction intermediates by rapid cooling to 100 K, and has traditionally been used to gain insight into long lived reaction intermediates such as product complexes. However, manual vitrification procedures are limited to long delay times of at least several seconds and heavily suffer from operator variability. A solution to this problem is provided by automatic crystal plunging devices, such as the Spitrobot, that plunge loop-mounted protein crystals into liquid nitrogen within millisecond time-scales. Versatile means of reaction initiation can be achieved either by micro dispensing a ligand droplet, or via optical excitation of light-sensitive proteins, or via the photoactivation of caged compounds. In addition to the conceptual simplicity, another benefit of cryo-trapping is that data can be collected at conventional synchrotron beamlines, exploiting their robust high-throughput capabilities. Thus, compared to room-temperature time-resolved crystallography, users not only benefit from uncoupling sample preparation and data-collection, but also from a reduction in the required technical expertise and ready access to radiation sources. However, as cryo-trapping crystallography explores dynamic structural changes that become only visible by the comparison of several samples, experiments have to be carefully planned to carry out the necessary controls and to avoid mis- or over-interpretation of the results. Here we describe a detailed protocol for cryo-trapping time-resolved crystallography using automated crystal-plungers that enables researchers to map enzymatic reaction coordinate pathways within the millisecond domain.