Modulation of liposome membranes by the C-terminal domain of the coronavirus envelope protein
Modulation of liposome membranes by the C-terminal domain of the coronavirus envelope protein
Alag, R.; Bui, M. H.; Miserez, A.; Torres, J.; Pervushin, K.; Sharma, B.
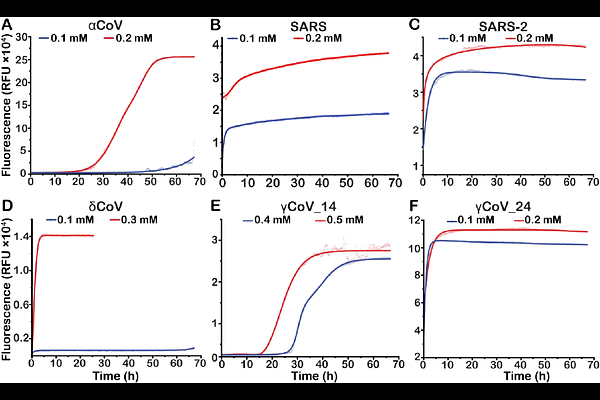
AbstractThe coronavirus envelope (E) protein is a viroporin that plays a key role in viral assembly, release, budding and pathogenesis. E protein forms oligomeric ion channels that can activate immune responses. However, high-resolution structural data for its extramembrane domains is limited. The C-terminal domain of SARS-CoV has been shown previously to form amyloid fibers, and here we show that these fibers can modulate the shape of liposomes. The propensity to form fibrils, and their effect on liposomes, was examined for sequences belonging to the four clades of coronaviruses. Electron microscopy data shows that the C-terminal domain in E protein adopts a filamentous structure. These findings demonstrate the potential of these peptides to modulate membranes, providing a possible mechanism by which E protein interacts with membranes in the host cell.