Developmental regulation of progenitor aging shapes long-term intestinal homeostasis in Drosophila
Developmental regulation of progenitor aging shapes long-term intestinal homeostasis in Drosophila
Malik, S.; Mahajan, A. A.; Pillai, S. J.; Shinde, I.; Shameem, M.; Chandrani, P.; Inamdar, M. M.; Khadilkar, R. J.
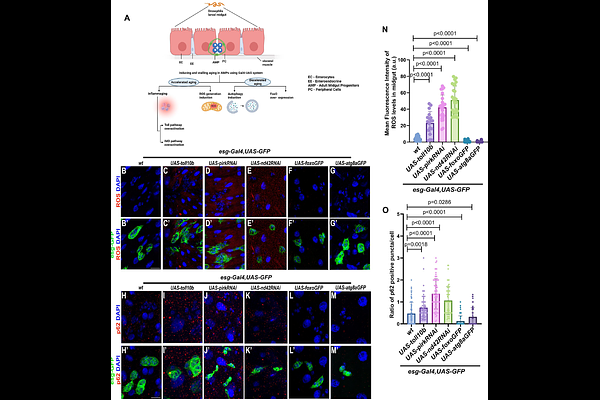
AbstractAging causes a progressive loss of tissue homeostasis, with stem cell exhaustion as a major hallmark. Age-associated decline in organ function is widely perceived as emanating from progressive accumulation of cellular damage in adult tissues. However, whether aging trajectories are established early on during development remains an open question. Here, we demonstrate that genetic modulation of cellular aging pathways in larval adult midgut progenitors (AMPs), which serve as the precursors of adult intestinal stem cells and differentiated epithelial cells, dictates the long-term trajectory of intestinal aging in Drosophila. Accelerated cellular aging by genetic perturbation employing Toll or Imd pathway overactivation or elevation of reactive oxygen species (ROS) using ND42 (mitochondrial complex I) knockdown in the AMPs results in aberrant progenitor proliferation, skewed lineage allocation, epithelial barrier dysfunction, and genomic instability. These alterations are accompanied by marked destabilization of AMP islet architecture and widespread changes in age-related molecular signatures, as revealed by bulk transcriptomic analysis. In contrast, decelerated cellular aging mediated by Foxo or Atg8a overexpression results in a decrease in enteroendocrine population and the intestinal barrier remained unaffected. Intriguingly, early-life activation of immune and oxidative stress signaling manifested later in the adult gut as elevated enteroendocrine differentiation, highlighting lasting effects on intestinal regenerative capacity and lineage balance. Together, our findings demonstrate that cellular aging is tightly regulated early on in development and its perturbation can cause developmental disruption hampering adult gut homeostasis, establishing AMPs as key developmental determinants that regulate the trajectory of intestinal aging in Drosophila.