Structural and biochemical characterization of a novel inhibitor of NMNAT1, the gatekeeper of nuclear NAD+ biosynthesis
Structural and biochemical characterization of a novel inhibitor of NMNAT1, the gatekeeper of nuclear NAD+ biosynthesis
Lansiquot, C.; Wu, R.; Davies, J.; Song, X.; Kaniskan, H.; Jin, J.; Lazarus, M. B.
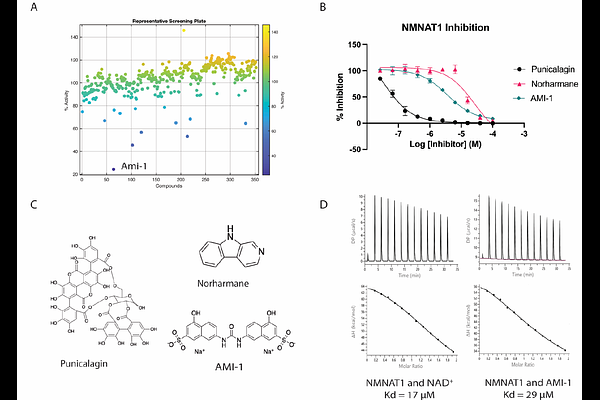
AbstractNicotinamide adenine dinucleotide (NAD+) is crucial for cellular functions including DNA repair and metabolism. Nicotinamide mononucleotide adenylyltransferase (NMNAT) enzymes catalyze the final step of NAD+ synthesis from NMN and ATP. There are three NMNAT isoforms: NMNAT1, NMNAT2, and NMNAT3, located in the nucleus, cytoplasm, and mitochondria, respectively. Nuclear NAD+ promotes disease progression in NAD+-dependent cancers, and it is hypothesized that targeting NMNAT1 with small-molecule inhibitors could be an effective therapeutic strategy. Here, we identify an NMNAT1 inhibitor from a bioactive compound screen and report its effects on NAD+ levels and the viability of NMNAT1-dependent cancer cell lines. The compound AMI-1 is a known inhibitor of Protein Arginine N-Methyltransferase 1, and we find that it also inhibits NMNAT1 with similar potency. Additionally, we determined a cryo-EM structure of NMNAT1 bound to AMI-1 and revealed its mechanism of inhibition. This provides proof of principle for inhibiting NMNAT1 to target NAD+ metabolism in dependent cancers, while also highlighting that caution is warranted when interpreting studies using AMI-1 as a PRMT1 inhibitor, given its effect on NAD+ through NMNAT1.