MaRNAV-1 infection of Plasmodium vivax is associated with increased parasite transmission and host inflammatory responses
MaRNAV-1 infection of Plasmodium vivax is associated with increased parasite transmission and host inflammatory responses
Seng, D.; Ko, K.; Orban, A.; Heng, S.; Feufack-Donfack, L.; Tebben, K.; Dumetz, F.; Grunebast, J.; Vignolini, T.; Dore, G.; Khim, N.; Salvador, J.; Ouaid, Z.; Lefevre, T.; Cohuet, A.; Sommen, C.; Flamand, C.; Ruberto, A.; Abebe, A.; Tsigie, M.; Malleret, B.; Shenkutie, T.; Lo, E.; Cantaert, T.; Anstey, N.; Petrone, M.; Baumgarten, S.; Serre, D.; Popovici, J.
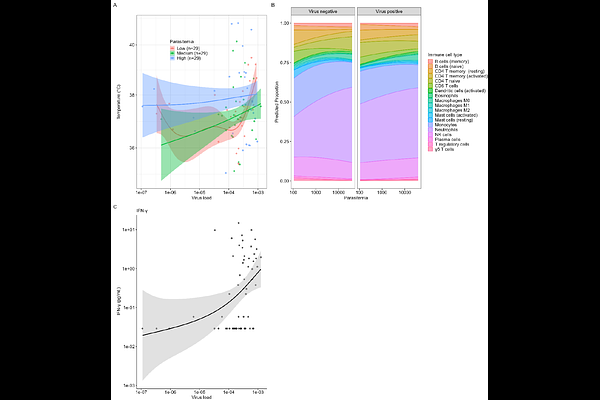
AbstractMatryoshka RNA virus 1 (MaRNAV-1) is an RNA virus recently identified in Plasmodium vivax infected samples, but definitive evidence that it infects the parasite and influences malaria pathogenesis remains unknown. Here, we demonstrate that MaRNAV-1 is an intracellular virus infecting P. vivax across various stages of its lifecycle, including blood stages, sporozoites, and liver stages. Viral prevalence varied geographically between Cambodia and Ethiopia, and sequence analyses revealed substantial nucleotide polymorphisms but a high level of protein conservation among viruses. MaRNAV-1 presence and load were positively associated with parasite transmission potential, as reflected by increased gametocyte abundance and higher oocyst prevalence and intensity in mosquito infection assays. In the patients, higher MaRNAV-1 loads were associated with elevated body temperature and increased concentrations of inflammatory cytokines, including IFN-{gamma}, IP-10, IL-6, IL-10, IL-1RA, and VEGF, independently of parasitemia. Consistent with this inflammatory profile, MaRNAV-1 loads were lower in asymptomatic infections compared to symptomatic malaria cases. Transcriptomic analyses further revealed differences in host immune-cell composition, including higher proportions of innate immune populations in virus-positive infections. Together, these findings demonstrate that MaRNAV-1 is a genuine parasite-infecting virus that contributes to the parasite's transmission potential and host's inflammatory response. Our study broadens the conventional view of host-pathogen interactions in malaria by revealing complex virus-parasite-host relationships.