Cell fate specification during respiratory development requires ARID1A-containing canonical BAF complex activity
Cell fate specification during respiratory development requires ARID1A-containing canonical BAF complex activity
Lee, H.; Jaquish, A.; Fernandes, S.; Zhao, B.; Elitz, A.; Cook, K.; Trovillion, S.; Arias, N. M. B.; Han, S.; Goodwin, S.; Russell, N.; Zacharias, A. L.; Brugmann, S. A.; Whitsett, J. A.; Sinner, D. I.; Sun, X. I.; Swarr, D. T.; Zacharias, W. J.
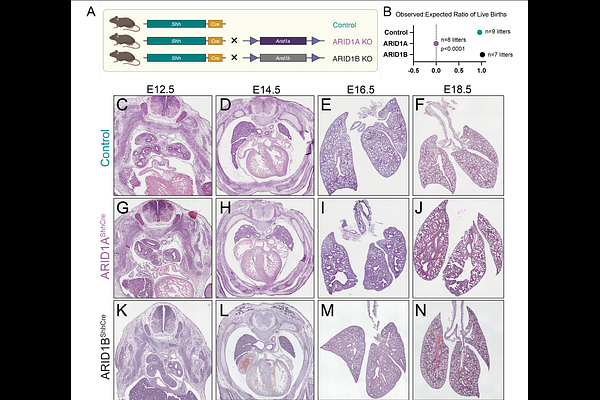
AbstractDevelopment of the mammalian lung requires formation of a definitive lung bud from the foregut endoderm, branching morphogenesis, specification of a proximal to distal axis, and differentiation of extensive specialized airway and alveolar epithelial lineages. These steps require coordinated temporal and regional gene expression to define the lung epithelium in preparation for the first breath at birth. While the transcriptional and signaling regulators required for lung development are increasingly well known, the key epigenetic complexes that interact with lineage transcription factors for cell-specific control of gene expression remain to be defined. Here, we identify a key role for the canonical BAF complex, an ATP-dependent chromatin remodeling complex, during lung epithelial development. Loss of canonical BAF activity throughout the foregut endoderm leads to complete failure of lung formation, and selective deletion of a single key subunit, ARID1A, leads to failure of proximal-distal axis specification, with dilated airway-like structures lined by ectopic basal cells found throughout the distal lung, and failure of specification of alveolar type 1 (AT1) cells in the distal saccular epithelium. In place of AT1 cells, we identified a highly proliferative epithelial cell state defined by joint activation of YAP and WNT signaling and loss of BMP signaling response, leading to increased proliferation and failure of appropriate epithelial differentiation. These changes resulted in secondary failure of mesenchymal and endothelial specification, leading to broad loss of patterning of the distal lung further disrupting peripheral lung morphogenesis. Using embryonic lung organoids, we demonstrate that exogenous BMP4 signaling is sufficient to rescue AT1 and AT2 cell differentiation in ARID1A-null epithelium, while WNT and YAP signaling require functional BAF complex. Together, these data demonstrate a requirement for the BAF complex for lung formation, proximal-distal patterning, and cell fate acquisition, and reveal surprising differential specificity between signaling pathways during lung development.