Herpes simplex virus pUL56 abolishes neuronal activity by removing voltage-gated ion channels from the plasma membrane
Herpes simplex virus pUL56 abolishes neuronal activity by removing voltage-gated ion channels from the plasma membrane
Nash, D. A.; Antrobus, P. R.; Nicholson, A. S.; Suberu, J.; Potts, M.; Andrada, M. A.; Lulla, V.; Crump, C. M.; Enright, A. J.; Weekes, M. P.; Deane, J. E.; Graham, S. C.
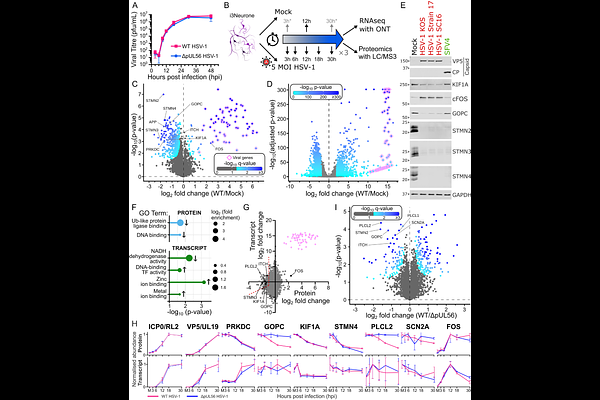
AbstractHerpes simplex virus 1 (HSV-1) infections of the central nervous system cause encephalitis and are associated with increased risk of neurodegeneration, yet the molecular consequences of lytic infection in human neurones remain incompletely defined. We map the transcriptomic, proteomic and surface-proteome changes induced by HSV-1 across the lytic infection cycle in human iPSC-derived cortical glutamatergic neurones. HSV-1 drives extensive plasma-membrane remodelling, including the removal of voltage-gated sodium, potassium and calcium channels, resulting in a profound loss of synchronous calcium signalling. We identify the viral ubiquitin-ligase adaptor pUL56 as the principal effector of this process: pUL56-dependent depletion of ion channels abolishes coordinated calcium signalling, whereas deletion or mutation of its E3-ligase binding motifs preserves synchrony. Furthermore, expression of pUL56 alone is sufficient to abolish neuronal electrical activity. These findings establish pUL56 as a potent viral suppressor of neuronal excitability and provide potential mechanistic links between HSV-1 infection and neurodegenerative pathology.