Auxin promotes GPI-anchored protein-mediated trafficking of ABP1 to enable cell-surface auxin signaling
Auxin promotes GPI-anchored protein-mediated trafficking of ABP1 to enable cell-surface auxin signaling
Wang, J.; Ye, J.; Zhang, M.; Feng, H.; Liu, M.; Huang, Y.; Xu, T.; Lu, B.; Li, C.
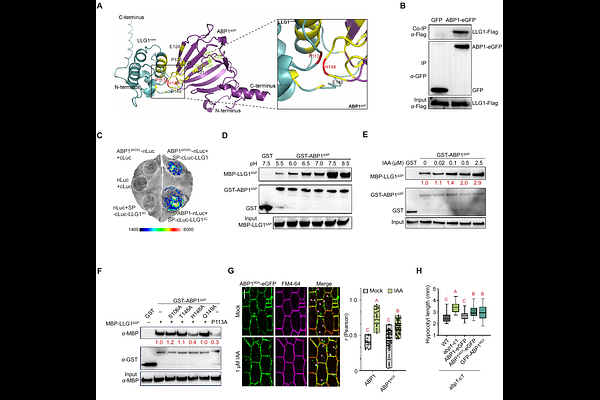
AbstractCell surface auxin signaling is essential for rapid cellular and growth responses to auxin. Auxin-Binding Protein 1 (ABP1) has been proposed as an extracellular auxin receptor, yet how the ER-retained ABP1 reaches the apoplast to function as an extracellular auxin receptor remains unclear. Here, we show that the abp1 mutants had strong phenotype in the dark-triggered hypocotyl elongation in Arabidopsis. More interestingly, the dark-induced auxin drives ABP1 relocation from the ER to the plasma membrane (PM) through a LORELEI-like GPI-anchored protein 1 (LLG1)-based mechanism. Auxin promotes LLG1 expression and its interaction with ABP1, facilitating ABP1 trafficking via a GPI-AP-specific pathway. Upon reaching the apoplast, ABP1 dissociates from LLG1 under acidic conditions, enabling its competence for auxin perception and activation of the cell surface signaling. Together, our results uncover a GPI-AP-mediated transport mechanism for ABP1 auxin receptor and refine the molecular framework of cell-surface auxin perception.