Did Iron Suppress Eukaryote Emergence and Early Radiation?
Did Iron Suppress Eukaryote Emergence and Early Radiation?
Adam, Z. R.; Slotznick, S.; Bauer, A. Z.; Stewart, E.; Roden, E.; Peters, S.; Konhauser, K.; Kacar, B.
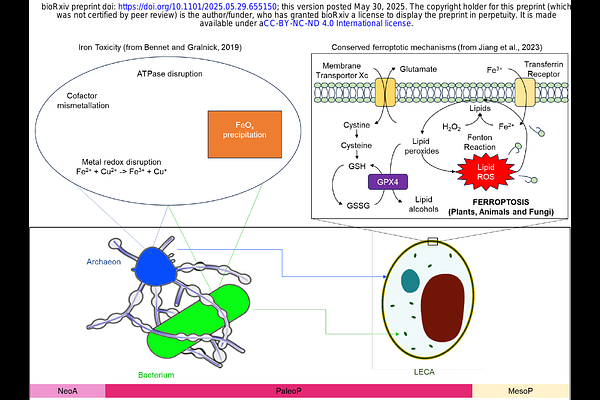
AbstractThe last eukaryotic common ancestor (LECA) is widely thought to have been an oxygen-respiring organism, arising through endosymbiosis when a free-living bacterium became the mitochondrion. Owing to the mitochondrion s central metabolic task of oxidative phosphorylation, oxygen availability has long been a hypothesized driver of eukaryogenesis. However, this hypothesis is challenged by a temporal disconnect, spanning several hundred million years, between the earliest geochemical evidence for oxygen in the environment (~3.2-2.5 Ga) and the oldest widely accepted eukaryotic fossils (~1.7 Ga). Notably, the earliest candidate eukaryotes appear contemporaneous with the cessation of major iron deposits and rise of sulfide- and sulfate-rich marine sediments in coastal environments. Here, we integrate Proterozoic surface geochemical records with the microbial biochemistry of iron to examine potential environmental constraints on early eukaryotic evolution. Iron bioavailability exerts complex and often antagonistic effects on both aerobic and anaerobic microbial lineages that contributed to LECA. Elevated iron levels likely disrupted cellular homeostasis, particularly by destabilizing labile iron pools and promoting oxidative damage to bacterial lipids. The programmed cell death pathways known as ferroptosis, which is widespread among eukaryotic lineages, may trace its origins to iron-rich conditions in Archaean and Paleoproterozoic seawater and LECA. Our findings challenge oxygen-centered paradigms of eukaryogenesis and reframes the long-recognized temporal gap as a consequence of iron-mediated physiological constraints.