Remifentanil self-administration promotes circuit- and sex-specific adaptations within the prefrontal-accumbens pathways
Remifentanil self-administration promotes circuit- and sex-specific adaptations within the prefrontal-accumbens pathways
Kokane, S. S.; Atwell, S. I.; Madayag, A. C.; Anderson, E. M.; Demis, S.; Engelhardt, A.; Friedrich, L.; Hearing, M. C.
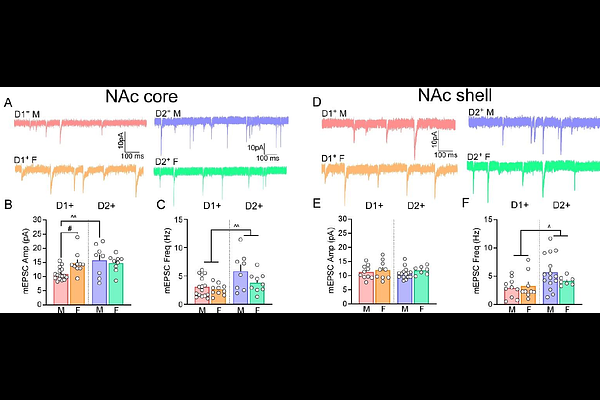
AbstractThe nucleus accumbens (NAc) and its excitatory input from the medial prefrontal cortex (mPFC) form a critical circuit underlying drug-induced plasticity associated with addiction-related behaviors. However, baseline differences in excitatory signaling across NAc subcircuits and sex-specific neuroadaptations following opioid self-administration remain poorly understood. Here, we examined synaptic signaling in mPFC-NAc pathways in drug-naive mice and after abstinence from remifentanil self-administration. Under drug-naive conditions, AMPA receptor-mediated glutamatergic signaling was generally elevated in D2 medium spiny neurons (MSNs) of both the NAc core and shell across sexes, while females exhibited greater excitatory signaling in D1 MSNs of the NAc core compared with males. Pathway-specific analyses revealed that prelimbic cortex (PL) inputs to NAc core D2 MSNs displayed enhanced calcium-permeable AMPA receptor (CP-AMPAR) signaling and increased presynaptic release relative to D1 MSNs. Following abstinence from remifentanil self-administration, miniature excitatory postsynaptic current analyses showed increased excitatory drive at D1 MSNs and decreased drive at D2 MSNs, largely restricted to the NAc core. At PL-Core D1 MSN synapses, remifentanil reduced AMPA/NMDA ratios, consistent with increased CP-AMPAR incorporation in males and females, while increasing presynaptic signaling exclusively in males. In contrast, PL-Core D2 MSN synapses showed a reduction in presynaptic signaling across sex, while ostensibly weakening postsynaptic signaling selectively in males through reductions in CP-AMPAR signaling. At infralimbic cortex (IL)-shell inputs, a reduction in AMPAR rectification indices at D1 MSN synapses was produced by remifentanil, while release probability was decreased at D2 MSN synapses in males only. Together, these findings reveal sex- and pathway-specific synaptic adaptations within mPFC-NAc circuits that may be obscured by global measures of excitatory transmission and identify baseline circuit differences that may shape opioid-induced plasticity.