Dynamic myosin 10 coupling to DCC and β1 integrin is mediated by intrinsically disordered regions during filopodial transport and patterning
Dynamic myosin 10 coupling to DCC and β1 integrin is mediated by intrinsically disordered regions during filopodial transport and patterning
Shangguan, J.; Reinhardt, S.; Weng, S. H. S.; Jungmann, R.; Sosnick, T. R.; Rock, R. S.
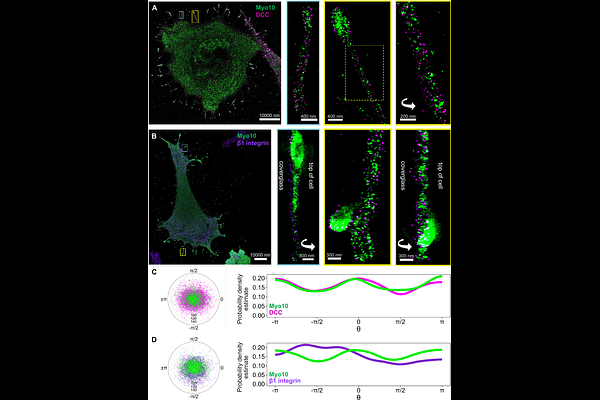
AbstractIntrinsically disordered regions (IDRs) are key mediators of protein-protein interactions. IDRs are important components of Myosin 10 (Myo10) and cargo complexes that influence neuronal development and cell growth, yet how IDRs dictate Myo10's cargo affinity and selectivity is not fully understood. Here, we investigate how the actin motor protein Myo10 engages two distinct cargo receptors, DCC and {beta}1 integrin, in cellular protrusions known as filopodia. Using hydrogen-deuterium exchange mass spectrometry (HDX-MS), cross-linking mass spectrometry (XL-MS), live-cell imaging, and super-resolution microscopy, we show that Myo10 decodes IDR elements through two complementary mechanisms: disorder-to-order transitions and "fuzzy" binding. The cytoplasmic portion of DCC binds Myo10 via a weakly helical P3 motif that acts as a preformed recognition element, while additional disordered motifs contribute to affinity through dynamic, weak interactions. In contrast, the {beta}1 integrin tail interacts with Myo10 through short NPxY motifs that remain disordered. Both cargos engage a common Myo10 surface but also contact distinct sites. Super-resolution DNA-PAINT imaging reveals distinct patterning of cargo with Myo10 along and around filopodia. Concentration measurements show that DCC is primarily bound while {beta}1 integrin exhibits a broader range of occupancy along the filopodial shaft. Multiple additive weak contacts and a shared binding site implies that DCC can out-compete integrin for Myo10 binding, which causes redistribution of active {beta}1 integrin from the filopodial tip to the shaft. Our findings illustrate a tunable, multivalent binding strategy that allows Myo10 to selectively coordinate diverse signaling cargos, demonstrating how regulated disorder within IDRs is one mechanism underlying cargo binding and cellular signaling.