Hierarchical decoding of targeting tripeptide motif by the cytosolic iron-sulfur cluster assembly targeting complex
Hierarchical decoding of targeting tripeptide motif by the cytosolic iron-sulfur cluster assembly targeting complex
Buzuk, A.; Khan, O.; Kang, S.; Yim, L.; Vajda, S.; Perlstein, D.
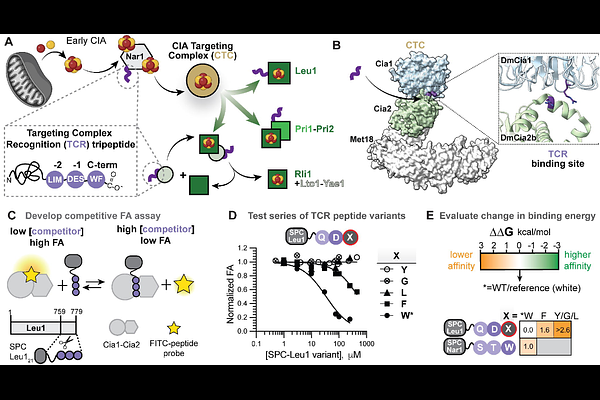
AbstractIron-sulfur (Fe-S) clusters are essential cofactors required for diverse cellular processes, yet how the Fe-S cluster biogenesis machinery selectively recognizes apo-client proteins remain poorly understood. In eukaryotes, many cytosolic and nuclear Fe-S proteins are recruited to the cytosolic iron-sulfur cluster assembly (CIA) system through a short C-terminal targeting complex recognition (TCR) motif having a [ILM]-[DES]-FW] consensus. Currently, the physicochemical properties underlying this molecular recognition event are undefined. By combining quantitative binding measurements, bioinformatic analysis, and structural modeling, we define the molecular basis for TCR peptide recognition by the CIA targeting complex (CTC). This systematic energetic dissection reveals a hierarchy of binding determinants, in which the side chain and C-terminal carboxylate of the aromatic residue provide the dominant energetic contributor, whereas the upstream residues modulate affinity in a sequence context-dependent manner. Computational docking and molecular dynamics simulations identify an interfacial binding site at the Cia1-Cia2 interface that can accommodate these TCR moieties complementary interaction surfaces. Mutational analysis the identified interaction site is consistent with an aromatic pocket and an adjacent hydrophobic groove on Cia2 accommodating the TCR's terminal aromatic and antepenultimate aliphatic residues. Together, these results reveal the physicochemical decoding grammar by which the CTC recognizes targeting peptides with divergent sequences, illustrating how short targeting motifs can achieve both the specificity and adaptability required for Fe-S protein maturation.