Lactate-dependent metabolic rewiring is associated with CD163+ macrophage polarization in human pyelonephritis
Lactate-dependent metabolic rewiring is associated with CD163+ macrophage polarization in human pyelonephritis
Borgards, L.; Voss, H. L.; Tautges, S.; Siebels, B.; Krisp, C.; Siemes, D.; Spangenberg, P.; Bottek, J.; Scharfenberg, S.; Schmitz, J.; Schwarz, L.; Rehme, C.; Hadaschik, B.; Braesen, J. H.; Roesch, A.; Schlueter, H.; Jorch, S.; von Vietinghoff, S.; Wagenlehner, F.; Shevchuk, O.; Engel, D. R.
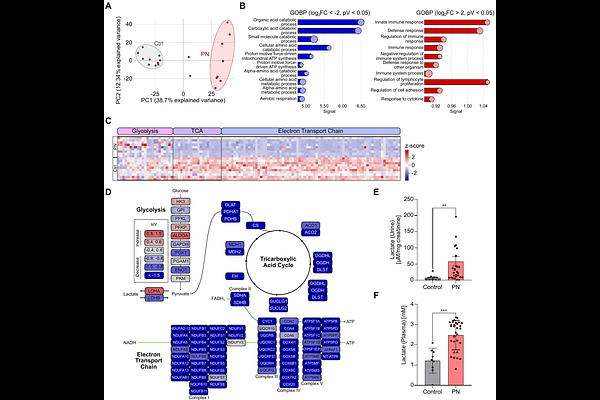
AbstractChronic pyelonephritis is characterized by persistent bacterial infection of the kidney and a dysregulated immune response that promotes disease progression. Macrophages are central regulators of antibacterial immunity in the urinary tract. However, the mechanisms underlying their functional reprogramming in chronic infection remain poorly understood. Here, we identify lactate as a key metabolic determinant of macrophage polarization in human pyelonephritis. Proteomic analysis of human kidney tissue revealed extensive metabolic remodelling, including upregulation of enzymes involved in glycolysis. Consistent with this, lactate levels were significantly elevated in urine and plasma of pyelonephritis patients. Concomitantly, we observed a pronounced accumulation of CD163 macrophages in infected kidneys, representing a distinct macrophage subset with immunomodulatory function. Correlation-based network analysis revealed a strong association between CD163 and lactate dehydrogenase A, supporting a functional association between lactate metabolism and macrophage polarization. Mechanistically, exposure of murine bone marrow-derived macrophages to lactate induced intracellular protein lactylation and promoted polarization toward a CD163 phenotype, defining a metabolically imprinted macrophage state distinct from classical activation paradigms. Proteomic profiling demonstrated extensive remodelling of macrophage protein expression in response to lactate with significant alteration of mediators of phagocytosis, Toll-like receptor signalling, and interferon response. Together, these findings identify lactate as a potent metabolic driver of macrophage reprogramming and establish a foundation for investigating lactylation-dependent immune dysfunction in chronic pyelonephritis.