A bivalent lysine-acetylated small-molecule binding site in MYC
A bivalent lysine-acetylated small-molecule binding site in MYC
Gupta, D. G.; Truica, M. I.; Steffeck, A. W. T.; Parker, J. B.; Ryan, D. H.; Pan, H.; Yang, W.; Lu, X.; Unno, K.; Quan, S.; Elmashae, A. A.; Kerber, M. M.; Brookins, C. E.; Han, H.; Dufficy, F.; Shim, J. g.; Lambert, M.; Ziarek, J. J.; Chakravarti, D.; Schiltz, G.; Abdulkadir, S. A.
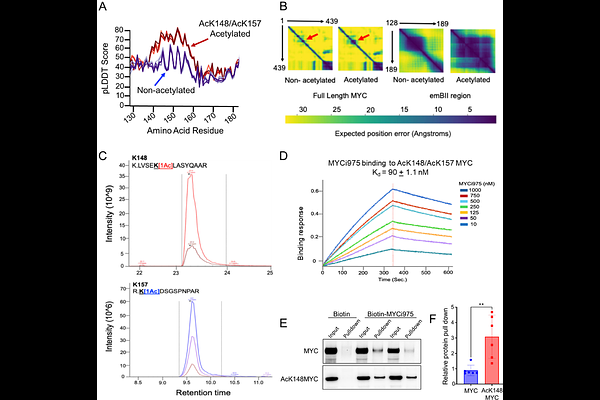
AbstractMYC is an important, yet challenging target in oncology as it lacks traditional druggable pockets. Here we show that two regions of the MYC protein, the basic-Helix-Loop-Helix (bHLH) domain, and an extended MYC Box II (eMBII) come together to form a bivalent, high-affinity small-molecule MYC inhibitor (MYCi) binding site. CRISPR-tiling mutagenesis identified mutations in the vicinity of emBII and in bHLH regions that together confer MYCi resistance. Importantly, acetylation of lysine K148 in eMBII which is essential for MYC oncogenicity in vivo increased the predicted order of this region and enhanced MYCi binding affinity. Furthermore, MYCi selectively modulated the expression of the same genes regulated by lysine-acetylated MYC in cancer cells. These studies provide a rationale for the selective targeting of acetylated, oncogenic MYC with small molecules.