Modulating Innate Immune Responses to Curli Fibers Through Protein Engineering
Modulating Innate Immune Responses to Curli Fibers Through Protein Engineering
Bonanno, S.; Sheta, R.; Ramu, T.; Verenkar, S.; Kim, D.; Bessette, E.; Pierre, P.; Joshi, N. S.
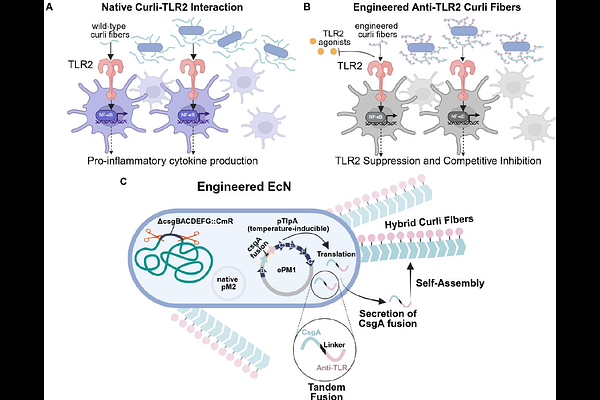
AbstractCurli fibers produced by Escherichia coli are functional amyloids that activate Toll-like receptor 2 (TLR2), initiating innate immune responses at mucosal surfaces. While microbiome-derived curli contribute to host-microbe interactions, their intrinsic immunostimulatory activity limits their utility as programmable scaffolds for engineered probiotic systems, and dysregulated TLR2 activation has been linked to inflammatory bowel disease, systemic lupus erythematosus, neurodegeneration, and sepsis. Here, we engineered E. coli Nissle 1917 to produce modified curli fibers designed to inhibit TLR2 through two mechanistically distinct strategies: steric shielding via silk-elastin-like protein sequences, and direct receptor antagonism via a known TLR2 antagonist, staphylococcal superantigen-like protein 3 (SSL3). Both engineered variants assembled into structurally intact amyloid fibers and exhibited significantly reduced intrinsic TLR2-dependent NF-{kappa}B activation in reporter cells. In competitive inhibition assays against structurally diverse TLR2 agonists, the SSL3 fusion achieved near-complete inhibition maintained under rising agonist load, while steric shielding provided moderate, agonist-class-dependent inhibition. In primary human monocyte-derived dendritic cells, the SSL3 fusion robustly attenuated IL-8 secretion and transcriptional induction of IL-8, IL-6, and IL-1{beta}, whereas steric shielding produced only partial attenuation that did not translate to broad inflammatory suppression. These results establish engineered curli as a tunable platform for receptor-specific modulation of innate immune signaling and highlight the broader potential of modular microbial amyloids as programmable interfaces for engineering host-microbe interactions at mucosal surfaces.