MicroRNA combinations function as synergistic network regulators of neuroblastoma differentiation
MicroRNA combinations function as synergistic network regulators of neuroblastoma differentiation
Lawson, S. A.; Zhang, Y.; Kosti, A.; Hart, M. J.; Penalva, L. O.; Pertsemlidis, A.
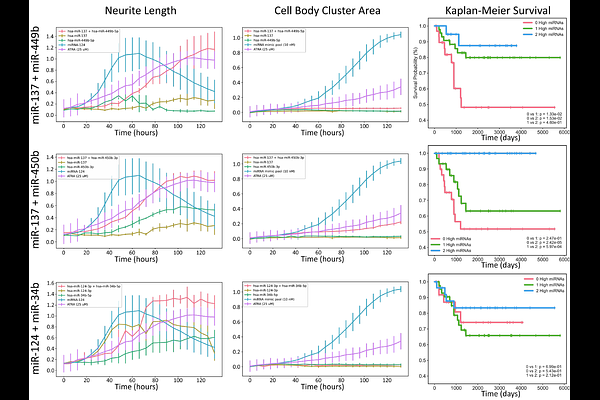
AbstractDifferentiation-based therapies represent a promising strategy for the treatment of neuroblastoma; however, single-agent approaches frequently yield incomplete and transient responses due to the robustness of underlying gene regulatory networks. MicroRNAs (miRNAs) are endogenous regulators of gene expression that modulate entire gene programs rather than individual molecular targets, making them attractive candidates for network-level therapeutic intervention. While individual miRNAs have been explored as therapeutic agents, the potential for synergistic interactions between miRNAs remains largely unexplored. Here, we developed a scalable high-content phenotypic screening platform to identify synergistic miRNA combinations that promote neuronal differentiation and growth arrest in neuroblastoma cells. Using SK-N-BE(2)-C cells and automated quantification of neurite outgrowth and confluence, we screened pairwise combinations of differentiation-associated miRNAs at submaximal doses. Candidate synergistic interactions were identified using the Highest Single Agent framework and subsequently validated by dose-response interaction modeling. We identified a robust synergistic interaction between miR-124-3p and miR-363-3p that exceeded zero-interaction potency expectations by approximately 20.9% and increased maximal differentiation-associated phenotypic response by 73% relative to single-miRNA treatments. Target gene and pathway enrichment analyses revealed that miR-124-3p and miR-363-3p regulate largely distinct but functionally complementary target gene sets. These complementary targets converged on neuronal differentiation and cell cycle control pathways, providing a mechanistic basis for their cooperative activity. Together, these findings establish miRNA combinations as programmable network regulators capable of inducing complex cellular phenotypes with greater efficacy than single agents. This work provides a conceptual and experimental framework for the rational discovery of synergistic miRNA therapeutics and suggests new avenues for differentiation-based treatment strategies in neuroblastoma and other diseases driven by dysregulated regulatory networks.