A lysyl oxidase (LOX)/bone morphogenetic protein-1 (BMP1) complex to facilitate collagen remodeling
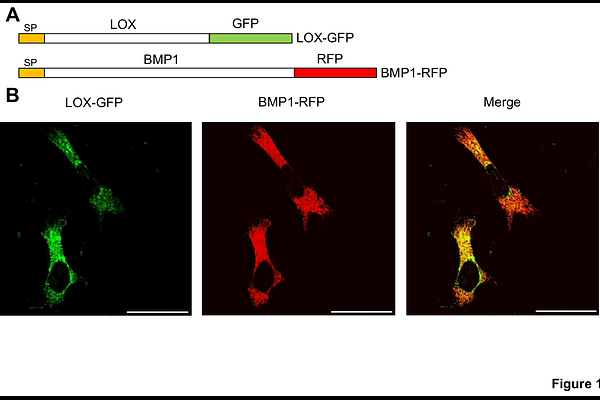
A lysyl oxidase (LOX)/bone morphogenetic protein-1 (BMP1) complex to facilitate collagen remodeling
Navarro-Gutierrez, M.; Romero-Albillo, V.; Rivas-Munoz, S.; Rosell-Garcia, T.; Jimenez-Sanchez, R.; Deen, M.; Poller, L. M.; Rodriguez-Pascual, F.
AbstractCollagen biosynthesis within the extracellular matrix (ECM) relies on finely regulated enzymatic steps to ensure proper collagen maturation and fibrillar assembly. Among these, bone morphogenetic protein 1 (BMP1) and the canonical lysyl oxidase (LOX) act on the collagen telopeptide to promote procollagen processing and oxidative cross-linking, respectively. However, the mechanisms that ensure precise coordination of their activities remain poorly understood. Using NanoBiT assays, we identified and characterized a stable LOX/BMP1 protein complex that assembles intracellularly during trafficking through the endoplasmatic reticulum (ER)/Golgi pathway and persists after secretion. Analysis of BMP1 and LOX domains involved in the interaction showed that BMP1 binding requires its CUB2/3 domains, while LOX recognition depends on a conserved, positively charged segment of LOX (residues 259-285) located immediately upstream of its catalytic domain. Formation of the LOX/BMP1 complex did not substantially alter LOX enzymatic activity but markedly enhanced LOX association with collagen type I through the carboxy-telopeptide region, facilitating the assembly of a ternary LOX/BMP1/collagen complex. This pre-assembled complex promoted efficient targeting of LOX to nascent collagen fibrils. Our findings reveal a previously unrecognized layer of regulation in collagen biosynthesis, in which LOX and BMP1 act as a functional unit to ensure precise localization and proper processing of collagen. This mechanism offers new insights into ECM assembly and identifies the LOX/BMP1 interface as a potentially druggable node for anti-fibrotic strategies.