Cyclome: Large-scale replica-exchange dynamics of 930 cyclic peptide reveal thermal stability and critical metal-binding behavior
Cyclome: Large-scale replica-exchange dynamics of 930 cyclic peptide reveal thermal stability and critical metal-binding behavior
Sajeevan, K. A.; Gates, H.; Raghunath, V. S.; Tan, C. P. H.; Danurdoro, R.; Young, J.; Chowdhury, R.
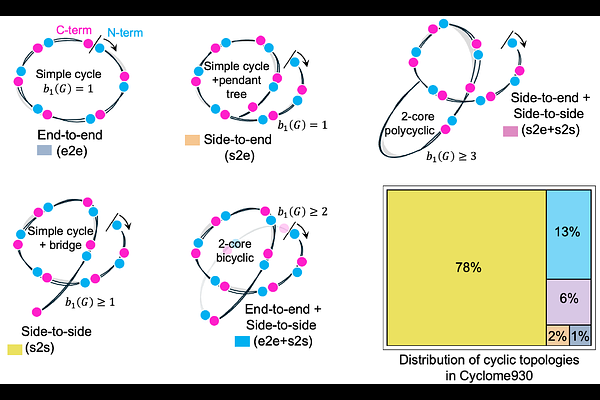
AbstractCyclic peptides are recognized as versatile scaffolds for therapeutic and functional applications due to their structural stability and resistance to degradation. Despite this promise, systematic analysis and prediction of their thermal stability remain limited by fragmented data resources, inadequate sequence comparison methods, and the lack of cyclicity-aware computational models. We provide a comprehensive, multi-scale computational framework to characterize cyclic peptides. First, we unified four fragmented public repositories of cyclic peptides into a single largest curated resource of 930 cyclic peptides, Cyclome930. This integrates cyclic topology, sequence, experimental structural coordinates, and source organism annotations into a consistently featurized dataset. Cyclome930 thus expands the dataset of annotated cyclic peptides by ~3.4 fold (from 276 to 930). Second, we developed a novel cyclic sequence alignment algorithm that explicitly accounts for rotational symmetry and knot topology, enabling more accurate scoring of sequence similarity than conventional linear alignments. Third, we investigate the thermal stability of cyclic peptides using extensive all-atom replica-exchange molecular dynamics (100ns; REMD) simulations, allowing conformational sampling across 298 K to 400 K and track its stress tensors with increasing temperature. Finally, these simulation-derived thermo-stability metrics were used to train a machine learning model to predict cyclic peptide melting points from sequence and topology (STop2Melt). Crucially, the model introduces cyclicity-aware embeddings derived from ESMc representations coupled with cyclic offset vector, capturing the peptides knot topology. STop2Melt achieved strong predictive performance on held-out peptides and outperforms baseline methods that neglect cyclic structure. Finally, we scored Cyclome930 (cyclic ligands) for critical mineral metal binding using a multi-classifier model (CritiCL). To our knowledge, Cyclome930 represents the first effort in peptide literature to integrate physics-based temperature ramped simulations, cyclic sequence similarity scoring, machine learning for thermal stability prediction and scoring them for critical metal binding. Cyclicity-aware computational toolchains (cyclome930.studio/) provide a foundational resource for computational design of stable cyclic peptide prototype libraries thereby annotating and expanding genomic islands linked to critical mineral recovery.