Dynamic optimization of extrachromosomal DNA copy number drives tumour evolution
Dynamic optimization of extrachromosomal DNA copy number drives tumour evolution
Gnanasekar, A.; Zhang, S.; Curtis, E. J.; Tang, J.; Shankar, V.; Wong, I. T.-L.; Yan, X.; Li, R.; Yang, L. M.; Yao, M. A.; Ventura, A.; Werner, B.; Chang, H. Y.; Mischel, P. S.
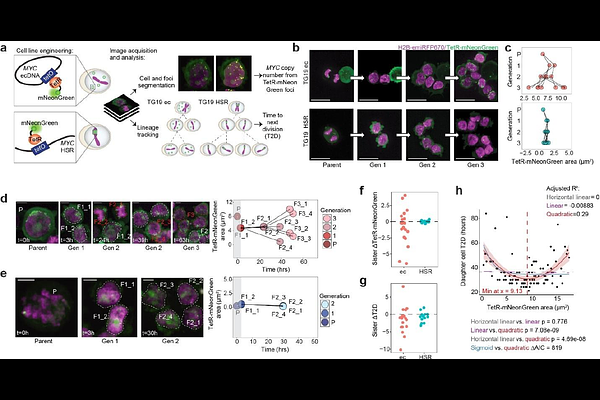
AbstractExtrachromosomal DNA (ecDNA) is common in human cancers and is associated with poor clinical outcomes, yet how ecDNA-driven genetic heterogeneity is translated into functional heterogeneity remains unclear. Using single-cell multiomics sequencing and multiplexed IF-FISH, we show that asymmetric inheritance of ecDNA generates copy number heterogeneity that propagates to gene expression programs, including oncogenic signaling and cellular stress responses. Transgenerational live-cell lineage tracking directly shows that ecDNA heterogeneity arises within only a few cell divisions and modulates daughter cell division timing in a copy number-dependent manner, a property not observed for evenly inherited chromosomal amplicons. We identify an optimal middle ecDNA copy number range that maximizes proliferative fitness at baseline, while drug selection pressure induced by low-dose CHK1 inhibition selects for cells with a new optimal range at low ecDNA copy numbers. These low ecDNA copy number cells pre-exist in the population and can be generated de novo, driving copy number shifts promoting drug resistance. In vivo experiments further demonstrate that shifts toward ecDNA copy numbers that are optimal under the tumour microenvironment enhance tumourigenicity. Together, these findings establish ecDNA copy number plasticity as a central driver of tumour evolution.