Viscoelasticity drives EMT in pancreatic intraepithelial neoplasia
Viscoelasticity drives EMT in pancreatic intraepithelial neoplasia
Pascucci, A.; Karim, S. A.; Morton, J. P.; Vassalli, M.; Walker, M. J.
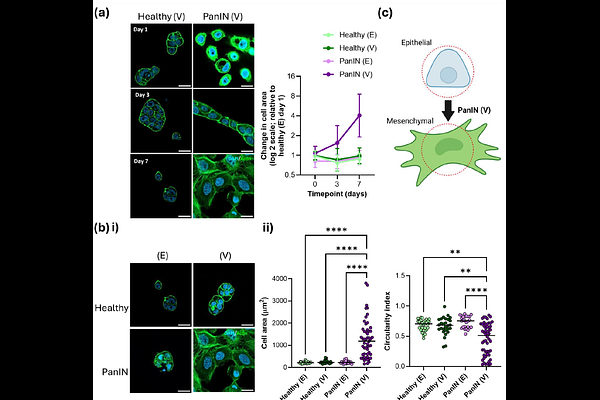
AbstractPancreatic intraepithelial neoplasia (PanIN) is a precursor of pancreatic adenocarcinoma (PDAC) and therefore critical to understand for identifying early-stage diagnostic and therapeutic targets. During PanIN, epithelial-to-mesenchymal transition (EMT) of pancreatic epithelial cancer cells is a crucial event which promotes invasion and early dissemination of cells into circulation before the full development of PDAC tumours. Changes in tissue mechanics are apparent during progression from PanIN to PDAC and increased local and global elasticity has been mathematically modelled in PanIN tissue as a predictive tool for diagnostics and development of personalized therapies. Aside from elasticity, viscoelasticity is emerging as a key feature of cancer which affects tissue mechanics through a combination of elastic and viscous components. Viscoelasticity has recently been shown to drive mechanosensitive cell behaviour and is known to change dramatically in PDAC progression. Hydrogels, as water-swollen polymer networks, are effective extracellular matrix (ECM) models that can recapitulate the viscoelastic properties of natural tissue. Despite this, hydrogels developed for studying cell behaviour in PanIN use purely elastic materials or have neglected the viscous component. Here, using PDAC mouse models, we show that viscoelasticity dynamically alters between healthy and PanIN-bearing tissue and have decoupled the role of elasticity and viscosity during EMT of pancreatic epithelial cancer cells using two-dimensional (2D) polyacrylamide (PAAm) hydrogels. Our work shows viscosity is critical in driving phenotypic changes associated with EMT in a pancreatic epithelial cancer cell line. These findings identify viscosity as an integral component of cell mechanosensing as PanIN develops, which may contribute to initial metastatic events via dissemination from the developing primary tumour. This should be explored further to potentially reveal novel diagnostic and therapeutic targets.